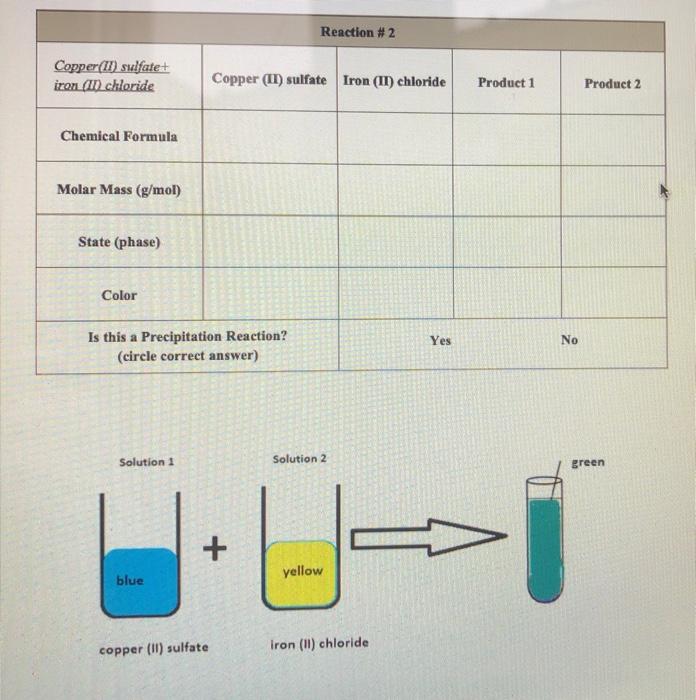
SOLVED:Consider reacting copper(II) sulfate with iron. Two possible reactions can occur, as represented by the following equations. copper(II) sulfate (a q)+ iron (s) ⟶copper (s) # iron(II) sulfate (a q) copper (

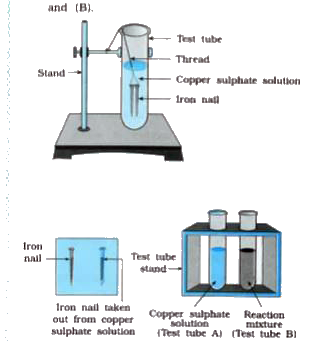
When an iron nail is placed in copper sulphate solution, we observe colour changes whereas when copper wire is placed in iron sulphate solution, we observe no change in colour. What is

Electron transfer reactions. A grey iron nail is dipped into blue copper sulfate solution and immediately withdrawn. Iron and copper sulfate. - ppt download

Class 9 Chemistry Practical's and Experiments on To carry out the reaction of Iron with copper sulphate solution in water and classify it as physical or chemical changes
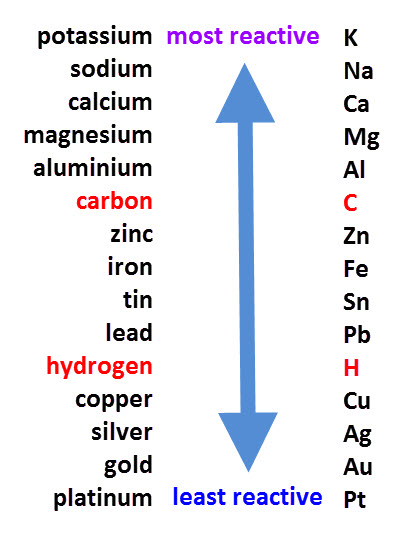
Chemical reaction takes place when iron dust is added into copper sulphate solution but no reaction takes place when copper dust is added into ferrous sulphate solution, why? | Socratic