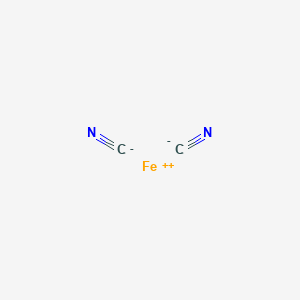
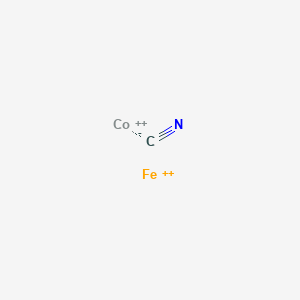SOLVED:Write the formula of (a) manganese(III) sulfide. (b) iron(II) cyanide. (c) potassium sulfide. (d) mercury(II) chloride.
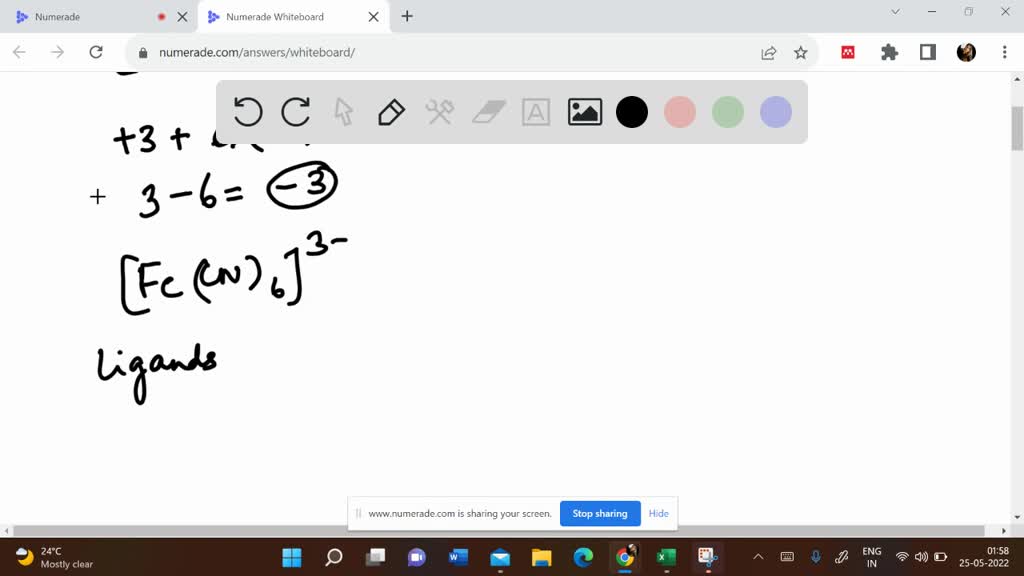
SOLVED: The iron(III) ion forms a complex with six cyanide ions that is often called the ferricyanide ion. What is the net charge on this complex ion, and what is its formula?
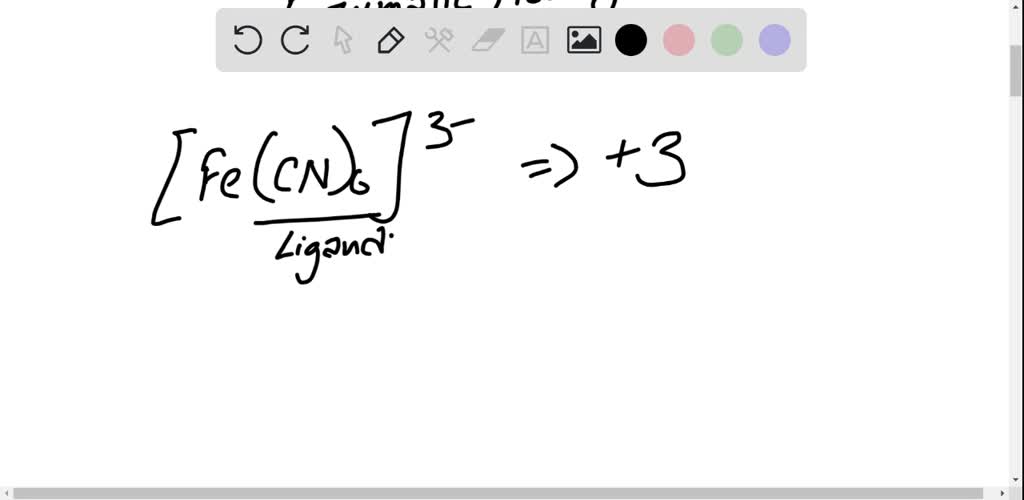
SOLVED:The iron(III) ion forms a complex with six cyanide ions that is often called the ferricyanide ion. What is the net charge on this complex ion, and what is its formula? What

SOLVED: 27. Choose the INCORRECT name formula combinationilmi (1 Point) LiCN lithium cyanide Fe2(S04)3 iron(III) sulfite Ba(OH)2 barium hydroxide HNO2 nitrous acid Na2co3 sodium carbonate

SOLVED: The formula for iron(II) cyanide is Fe(CN)2 How many grams of iron( II) cyanide are present in 4.18 X 1022 formula units of this compound? grams How many formula units of iron(ID)